The core mechanistic difference: semaglutide acts on the GLP-1 receptor, while tirzepatide activates both GIP and GLP-1 receptors. This dual action may contribute to stronger effects on appetite regulation, blood sugar control, and weight loss.
Two medications now dominate the clinical conversation on weight management: tirzepatide, prescribed under the brand name Mounjaro in the UK, and semaglutide, prescribed as Wegovy. Both belong to a class of weekly injection treatments that work with the body’s appetite-regulating hormones. Both have produced results that dwarf anything the previous generation of weight loss medications could achieve.
But they are not the same drug. They work through different mechanisms, produce different outcomes in clinical trials, carry subtly different side effect profiles, and suit different patients depending on their medical history and priorities.
This article compares the evidence on both, drawing from the major clinical trials including the first direct head-to-head study published in the New England Journal of Medicine, to give clinicians and patients a clear picture of where each medication stands.
How each drug works: the mechanism
Semaglutide: single GLP-1 agonism
Semaglutide is a glucagon-like peptide-1 (GLP-1) receptor agonist. It mimics the action of the naturally occurring GLP-1 hormone, which your gut produces after eating. By activating GLP-1 receptors in the hypothalamus and brainstem, it suppresses appetite, slows gastric emptying, stimulates insulin secretion, and reduces glucagon release.
Semaglutide was originally developed as a treatment for type 2 diabetes under the brand name Ozempic. The same molecule, reformulated at a higher dose of 2.4mg weekly, is what is approved for weight management as Wegovy. These are different formulations of the same drug, and the distinction matters: Wegovy is approved for obesity; Ozempic is approved for diabetes. Prescribing Ozempic off-label for weight loss is common but not the same as being prescribed Wegovy.
Tirzepatide: dual GLP-1 and GIP agonism
Tirzepatide is a dual agonist, meaning it activates two receptors simultaneously: GLP-1 and GIP (glucose-dependent insulinotropic polypeptide). GIP is a separate incretin hormone that plays its own role in regulating insulin secretion, fat metabolism, and energy storage. In isolation, GIP actually promotes fat storage. The interaction between GIP and GLP-1 receptor activation appears to flip this, producing additive or even synergistic effects on appetite suppression and fat metabolism that exceed what GLP-1 activation alone achieves.
Tirzepatide is the molecule behind Mounjaro, which was originally approved for type 2 diabetes and later approved in the UK by the MHRA for weight management under the NICE technology appraisal TA1026 in 2024. The mechanism is why it is sometimes called a “twincretin,” and the dual pathway activation is the primary reason it outperforms semaglutide in head-to-head clinical data.
Trial outcomes: what the data shows
Individual trial results
The STEP-1 trial for semaglutide enrolled adults with obesity but without type 2 diabetes and showed an average body weight reduction of 14.9% over 68 weeks at the 2.4mg dose. Approximately 86% of participants achieved at least 5% weight loss.
The SURMOUNT-1 trial for tirzepatide showed an average body weight reduction of up to 22.5% over 72 weeks at the 15mg dose in adults without type 2 diabetes. At the maximum dose, 91% of participants achieved at least 5% body weight loss, with 57% achieving at least 20%.
Comparing individual trials is inherently limited because they enrolled slightly different populations, ran for different durations, and used different measurement conventions. These figures suggest tirzepatide outperforms, but cross-trial comparisons are not definitive evidence.
SURMOUNT-5: the first direct comparison
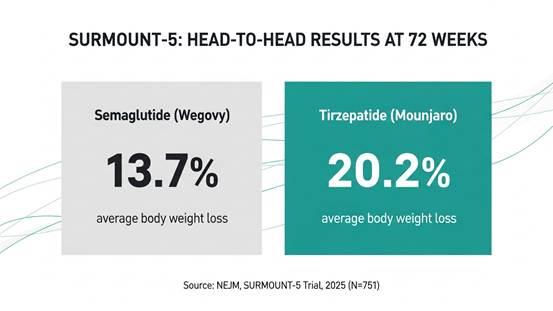
The definitive answer came in SURMOUNT-5, published in the New England Journal of Medicine in May 2025. This was the first phase 3b, head-to-head, randomised trial directly comparing tirzepatide and semaglutide in the same population. 751 adults with obesity but without type 2 diabetes were assigned 1:1 to the maximum tolerated dose of tirzepatide (10mg or 15mg) or semaglutide (1.7mg or 2.4mg) for 72 weeks.
SURMOUNT-5, published in the New England Journal of Medicine, May 2025: tirzepatide produced 20.2% average body weight loss vs 13.7% for semaglutide over 72 weeks. (N=751, adults with obesity without type 2 diabetes.)
The results were unambiguous. Tirzepatide produced 20.2% average body weight loss versus 13.7% for semaglutide, a difference of 6.5 percentage points and a 47% greater relative weight loss.
The milestone weight loss thresholds showed the same pattern. 51.6% of tirzepatide participants achieved at least 20% body weight loss, compared to 31.5% on semaglutide. At the 25% threshold, 31.6% of tirzepatide participants achieved this versus 16.1% on semaglutide.
Waist circumference reductions also favoured tirzepatide, with a mean reduction of 18.4cm versus 13.0cm on semaglutide.
|
“ |
In SURMOUNT-5, tirzepatide produced 20.2% average body weight loss vs 13.7% for semaglutide at 72 weeks. 31.6% of tirzepatide participants achieved 25% or more body weight loss, compared to 16.1% on semaglutide. Aronne et al., New England Journal of Medicine, SURMOUNT-5, May 2025 (N=751). Funded by Eli Lilly. |
Real-world evidence
A large retrospective cohort study published in Diabetes, Obesity and Metabolism in 2025, using Truveta de-identified US electronic health records (N=18,000+), confirmed the trial pattern in clinical practice: tirzepatide was associated with 15.3% body weight loss over 12 months versus 8.3% for semaglutide in patients without diabetes. The gap narrowed in patients with type 2 diabetes, where both medications performed more similarly.
A systematic review and meta-analysis published in 2025, covering seven studies totalling nearly 29,000 participants, also found tirzepatide consistently produced greater weight loss across both randomised controlled trials and observational studies.
Metabolic and cardiovascular outcomes
Metabolic markers
Both medications improve key metabolic markers alongside weight loss. Both reduce HbA1c, fasting insulin, triglycerides, blood pressure, and improve HDL cholesterol.
In SURMOUNT-5, tirzepatide demonstrated significantly greater improvements than semaglutide in systolic and diastolic blood pressure, HbA1c, fasting insulin, triglycerides, and HDL cholesterol. HDL increased by 7 to 8% with tirzepatide compared to approximately 4% with semaglutide.
A post-hoc cardiovascular risk analysis of SURMOUNT-5, published in European Heart Journal Open, found tirzepatide was associated with a greater reduction in predicted 10-year cardiovascular disease risk: an absolute reduction of 2.4% from baseline versus 1.4% for semaglutide.
Cardiovascular outcome data
On dedicated cardiovascular outcomes data, the picture is more nuanced. Semaglutide currently has the more mature evidence base. The SELECT trial demonstrated that semaglutide reduced major adverse cardiovascular events (MACE) by 20% in people with established cardiovascular disease and obesity, making it the first anti-obesity medication to demonstrate cardiovascular benefit in a dedicated outcomes trial.
Tirzepatide’s dedicated cardiovascular outcomes data comes from the SURPASS-CVOT trial, which demonstrated non-inferiority to insulin degludec for cardiovascular safety. However, a dedicated trial showing cardiovascular benefit (equivalent to SELECT) has not yet been published. This is an important distinction for patients with established cardiovascular disease: semaglutide currently carries stronger dedicated outcomes evidence for cardiovascular risk reduction. For patients whose primary goal is weight loss and broader metabolic health, rather than secondary cardiovascular prevention, tirzepatide has the stronger evidence.
Side effects: a detailed comparison
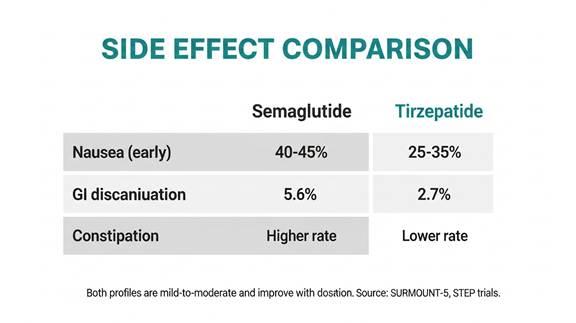
Side effect comparison from SURMOUNT-5 and STEP trial data. Both profiles are mild to moderate and improve significantly over time with proper dose titration and clinical monitoring.
Gastrointestinal effects
Both medications produce gastrointestinal side effects, primarily nausea, diarrhoea, vomiting, and constipation. These are largely a consequence of slowed gastric emptying and central nervous system effects that both drugs produce. They are most common during the dose escalation phase and typically improve once a stable dose is reached.
Nausea rates across trial data run at approximately 40 to 45% for semaglutide at therapeutic doses and 25 to 35% for tirzepatide, depending on dose level. However, cross-trial comparisons of side effect rates should be interpreted carefully due to differences in reporting methodology.
The SURMOUNT-5 head-to-head data provides the most reliable comparison. Gastrointestinal adverse events causing treatment discontinuation occurred in 5.6% of semaglutide participants versus 2.7% of tirzepatide participants, suggesting tirzepatide is somewhat better tolerated despite producing greater weight loss.
Constipation rates are also higher with semaglutide at the 2.4mg dose than with tirzepatide at comparable stages of treatment. Tirzepatide does show slightly higher rates of certain GI symptoms at very high doses, but the net tolerability data from the head-to-head trial favours tirzepatide.
Muscle mass preservation
An often-overlooked difference concerns lean mass preservation. DEXA substudy data from STEP-1 showed that approximately 45% of total weight lost on semaglutide at 2.4mg came from lean mass. SURMOUNT-1 body composition data for tirzepatide showed approximately 26% of total weight lost came from lean mass. Both figures reflect the importance of resistance training and adequate protein intake during treatment, but tirzepatide appears to produce a meaningfully better fat-to-lean mass ratio at the doses studied.
Dosing and escalation
Both medications follow a dose escalation schedule to minimise side effects. Semaglutide (Wegovy) starts at 0.25mg weekly and escalates through 0.5mg, 1mg, 1.7mg, and finally 2.4mg over approximately 16 weeks.
Tirzepatide (Mounjaro) starts at 2.5mg weekly and escalates through 5mg, 7.5mg, 10mg, 12.5mg, and up to 15mg, typically increasing every four weeks. The escalation schedule is longer, which means reaching maximum dose takes more time, but it also gives the body more adjustment steps at each level.
The prescribing decision
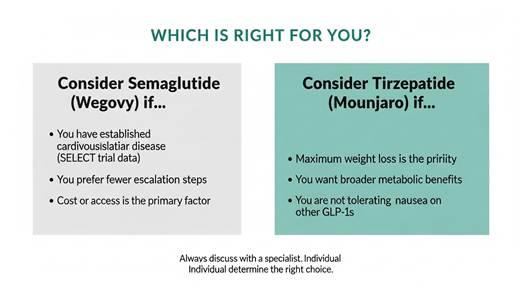
Clinical decision framework. Individual factors, including cardiovascular history, metabolic goals, tolerability, and access, should all inform the prescribing conversation between patient and specialist.
For most patients whose primary goal is maximum weight loss and broad metabolic improvement, the evidence from SURMOUNT-5 points clearly toward tirzepatide. It produces greater body weight reduction, better metabolic marker improvements, superior lean mass preservation, and lower rates of GI-driven discontinuation.
Two factors complicate this straightforward conclusion.
The first is cardiovascular history. If a patient has established cardiovascular disease and the treatment goal includes reducing future cardiovascular events, semaglutide currently carries stronger dedicated outcomes evidence (SELECT trial). This does not mean tirzepatide is cardiovascularly unsafe. It means the evidence base is less mature for dedicated cardiovascular risk reduction specifically, and clinicians should factor this in for high-risk patients.
The second is tolerability and individual response. Despite tirzepatide’s better average profile, individual variation is substantial. Some patients tolerate one medication better than the other, and prescribing decisions should account for prior GI history, comorbidities, and patient preference alongside the trial data.
Accessing tirzepatide through a supervised programme
For patients who, after reviewing the evidence, want to explore tirzepatide as a weight loss treatment, Voy provides access to supervised tirzepatide treatment following an online assessment and review by a UK-registered prescriber. The programme includes eligibility checks, dose planning, delivery after approval, and ongoing clinical support. 88% of Voy members are on a bespoke, non-standard plan, reflecting the clinical reality that one prescribing approach does not fit all patients.
|
“ |
Voy members on supervised tirzepatide treatment achieved 24% average weight loss at 12 months, outperforming the 16% benchmark from clinical trials, with 100% quality-of-life improvement and 100% reversal of pre-diabetes HbA1c. Published in JMIR, 2025 (N=125,000+). Individual results may vary. Treatment is subject to clinical suitability. |
Summary: where the evidence stands
On the question of which medication produces greater weight loss, the first direct clinical trial has now provided a definitive answer: tirzepatide outperforms semaglutide at maximum tolerated doses, with 20.2% versus 13.7% average body weight loss at 72 weeks in the SURMOUNT-5 trial.
On broader metabolic outcomes, tirzepatide again shows advantages: better blood pressure reduction, better cholesterol improvement, better HbA1c reduction, and superior lean mass preservation.
On cardiovascular outcomes specifically in patients with established cardiovascular disease, semaglutide currently has the stronger dedicated evidence from the SELECT trial. Tirzepatide’s cardiovascular outcomes data is less mature for this specific indication.
Both medications represent a genuine step change in what is achievable through pharmacological weight management. The question of which to prescribe is increasingly one of patient-specific factors rather than fundamental efficacy, since both produce outcomes that were impossible with any previous treatment option.
|
This article is for informational purposes and does not constitute medical advice. Always consult a healthcare professional before starting any weight loss treatment. Individual results may vary. Treatment is subject to clinical suitability. Results achieved alongside a reduced-calorie diet and increased physical activity. |
Sources
• Aronne LJ et al. Tirzepatide as Compared with Semaglutide for the Treatment of Obesity. SURMOUNT-5. New England Journal of Medicine, May 2025.
• Jastreboff AM et al. Tirzepatide Once Weekly for the Treatment of Obesity. SURMOUNT-1. New England Journal of Medicine, 2022.
• Wilding JPH et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. STEP-1. New England Journal of Medicine, 2021.
• Lincoff AM et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. SELECT. New England Journal of Medicine, 2023.
• Mamas MA et al. Tirzepatide compared with semaglutide and 10-year cardiovascular disease risk reduction in obesity: SURMOUNT-5 post-hoc analysis. European Heart Journal Open, 2025.
• Ghusn W et al. Real-world effectiveness of tirzepatide vs semaglutide for weight loss. Diabetes, Obesity and Metabolism, 2025.
• Systematic review and meta-analysis: Tirzepatide vs Semaglutide, 29,000 participants. PubMed, 2025.
• Voy outcome data: JMIR, 2025 (N=125,000+). Diabetes Obesity and Metabolism, 2025. Cureus, 2025.